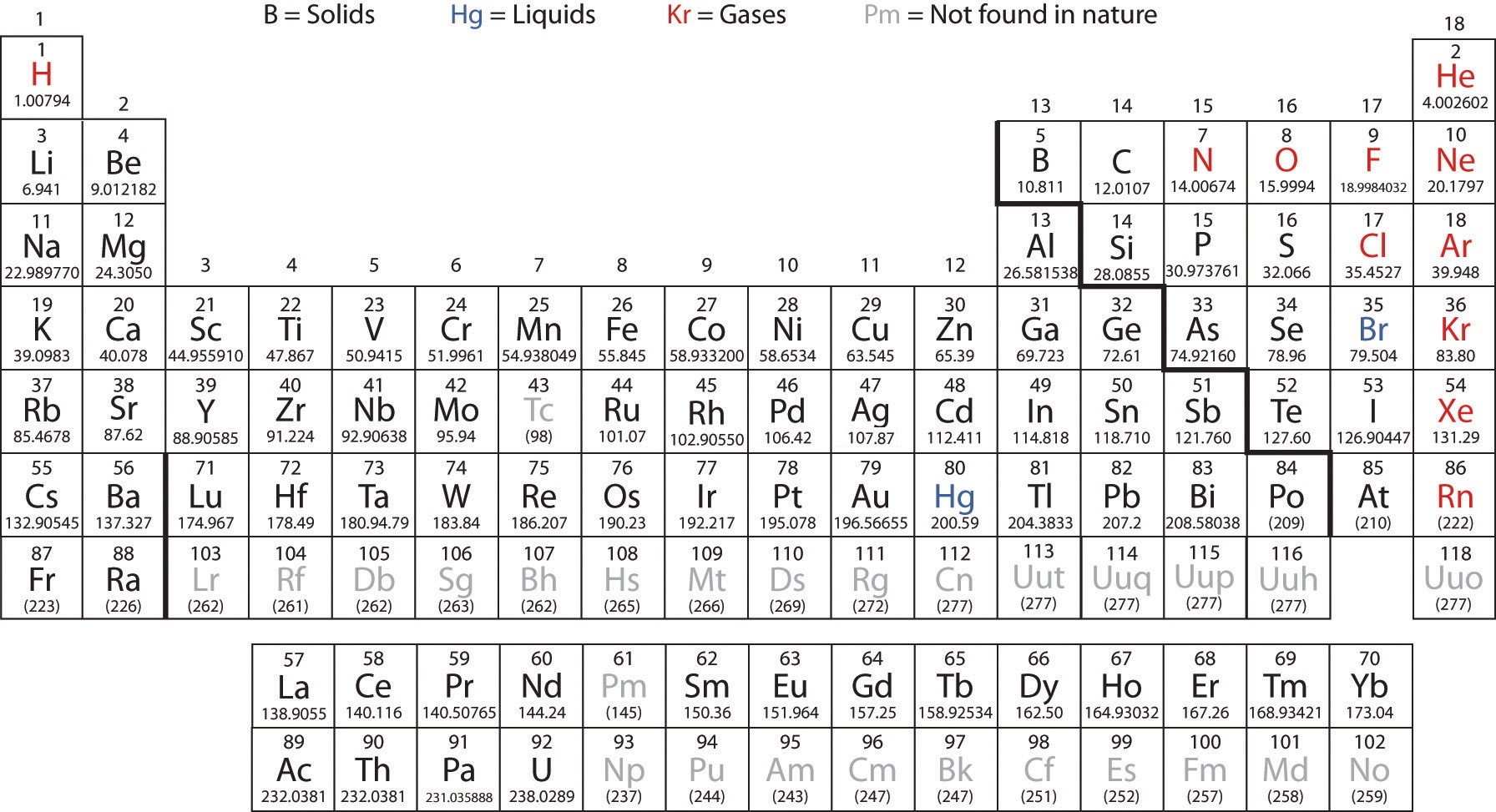
  He kept his discovery secret until 1680, when Anglo-Irish chemist Robert Boyle rediscovered phosphorus and published his findings. In 1669, or later, his experiments with distilled human urine resulted in the production of a glowing white substance, which he called "cold fire" ( kaltes Feuer). Brand tried to discover the philosopher's stone-a mythical object that was supposed to turn inexpensive base metals into gold. The first person in recorded history to discover a new element was Hennig Brand, a bankrupt German merchant. The history of the periodic table is also a history of the discovery of the chemical elements. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Hennig Brand, as shown in The Alchemist Discovering Phosphorus Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). WHO ARRANGED THE ELEMENTS ACCORDING TO ATOMIC MASS FREELet me know in the comments below, whether this article helped you or not? Free Gift for you: Interactive Periodic Table I hope you have clearly understood the arrangement of elements in Mendeleev’s First Periodic table. The Noble gases were not discovered during Mendeleev’s time, so they were not included in his table. The elements lying in the same column were having similar properties. Later on, his prediction was proved completely true when those three elements were discovered. The genius work which Mendeleev did was to leave empty gaps for the three elements whose properties were not matching. The elements in this First Periodic table were arranged in the increasing order of their atomic masses, and there were only 63 elements in his table. Mendeleev’s first periodic table published in 1869 was a huge success for the further development of Periodic table. This was also the great achievement of Mendeleev’s periodic table. In Mendeleev’s table, noble gases were not present.īut they can be placed in the separate column without disturbing the position of any elements. Now what about the position of Noble gases in Mendeleev’s table? Hence they were not discovered till 1869 during the time of Mendeleev. So they were not found with any compounds existing in nature.Īlso the noble gases are present in very few amounts in the atmosphere. What about the arrangement of Noble gases?ĭuring Mendeleev’s time, the noble gases were not discovered.īut do you know why noble gases were not discovered during that time?Īnswer: Noble gases are chemically inert. So Mendeleev’s periodic table had few exceptions in which the increasing order of the atomic mass was not followed. Let me give you an example from his table. The simple answer: Mendeleev placed some elements with higher atomic mass before lower ones, so that he can group the elements with similar properties together. He placed some elements with greater atomic mass before the elements with lower atomic mass. Exceptions in arrangement of Mendeleev’s periodic table That is why Mendeleev is also known as Father of Periodic table. He also predicted the properties of these three elements and also stated that they will be discovered in future.Īfter few years, these 3 elements were discovered as Īnd the main thing is that the properties of these three elements were almost similar to the properties that Mendeleev had already predicted. He predicted that those three elements were not discovered and he named those three elements as eka-boron, eka-aluminium and eka-silicon. But instead of that, he left the gaps for the elements which were not showing the similar properties. Mendeleev didn’t try to fit all the elements forcefully in his table. The modified version of Mendeleev’s Periodic table is as shown below. In this way, Mendeleev prepared his first Periodic table in 1869. And if the property of elements didn’t match, then he placed it in the row. If the property of the elements were similar, then he used to place it in the same column. He started arranging these elements in the increasing order of their atomic masses from left to right in a row. 
Mendeleev prepared the cards of all the 63 known elements with their properties written on them. So according to Mendeleev, these elements should lie in the same column in his table. Hence they have similar chemical properties and they can be grouped together. Potassium reacts with hydrogen and oxygen to form KH and K 2O.įrom the above two examples we can easily say that both sodium and potassium form similar compounds having formulae RH and R 2O. Sodium reacts with hydrogen to form sodium hydride (NaH) and it reacts with oxygen to form sodium oxide (Na 2O). Why hydrogen and oxygen? Why not any other gas?īecause hydrogen and oxygen are the gases which react with almost all the elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
